Intranasal Insulin for TBI
Using the intranasal (IN) delivery method to target insulin to the central nervous system (CNS) originally developed by Frey, Born and coworkers demonstrated that cerebrospinal fluid insulin levels significantly increased after treatment of normal adults with insulin, with no change in blood levels of insulin. In normal adults, IN treatment with insulin for 8 weeks improved memory (delayed recall of words) and mood at doses that did not alter blood levels of insulin or glucose.
The sum of insulin’s effects on the brain include the following:
- improves brain ATP production
- decreases CSF cortisol
- improves neuronal viability in the hippocampus
- increases the expression of anti-inflammatory microglia
- reduces beta-amyloid and tau protein deposition
Intranasal (IN) insulin has extraordinary effects on the brain, now visualized and proven. See below figures for the effects on the neurons of the hippocampus and the microglia.
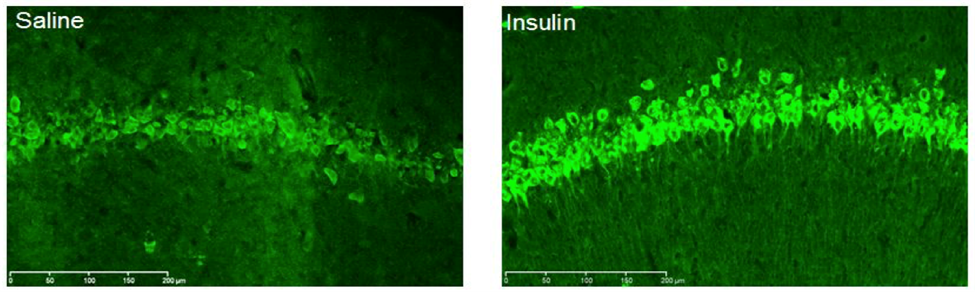
Figure 2. NeuN staining was increased with intranasal insulin treatment
TBI results in neuronal cell death. Neuronal cell death in the hippocampus impairs memory function. NeuN, an immunohistochemical marker of neurons, was used to examine the effect of intranasal insulin on neurons after injury. Qualitative assessment of histology showed improved neuronal viability in the hippocampus of the insulin treated rats.
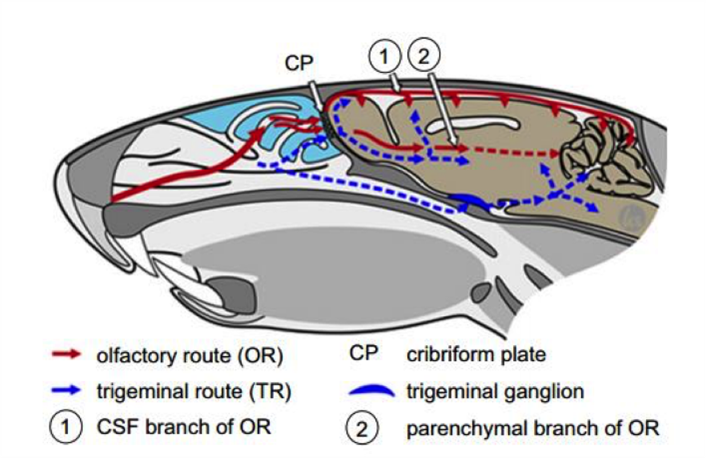
How Does Intranasal Insulin Reach the Brain?
Using the Olfactory Nerve channels to bypass the blood brain barrier with intranasal insulin, the following diagram is key for understanding how substances can get into the brain—in the CSF or parenchyma. One question commonly asked is, how long does it take for substances to get into the brain via this intranasal delivery? The consistent answer provided by the researchers and the studies is 10-15 minutes for the substances to be detected in the CSF after intranasal administration.
Why is Insulin Administered Intranasally?
Safety and Side Effects
Bloody noses and swallowing plasma or local anesthesia have been the biggest side-effects of IN PRP. If the patient swallows the insulin, he or she may need some form of sweet substance to eat to prevent a drop in blood sugar. Sneezing is another danger of the treatment. Using good sterile technique will prevent infection.
“The only unpleasant side effect of intranasal Humulin R is the distaste of it and ingestion by swallowing. As we know, the stomach acids rapidly denature the proteins that make up insulin such that any effects of the drug are eliminated such that blood sugar will not be affected. (This is why insulin is regularly injected by diabetics because no oral insulin can work).”
Scientific References
Bypassing the Blood Brain Barrier
Born, J., Lange, T., Kern, W., McGregor, G. P., Bickel, U., & Fehm, H. L. (2002). Sniffing neuropeptides: a transnasal approach to the human brain. Nature neuroscience, 5(6), 514-516.
Hanson, L. R., & Frey, W. H. (2008). Intranasal delivery bypasses the blood-brain barrier to target therapeutic agents to the central nervous system and treat neurodegenerative disease. BMC neuroscience, 9(Suppl 3), S5.
For Alzheimers
Craft, S., Baker, L. D., Montine, T. J., Minoshima, S., Watson, G. S., Claxton, A., … & Green, P. S. (2012). Intranasal Insulin Therapy for Alzheimer Disease and Amnestic Mild Cognitive Impairment. Archives of neurology, 69(1), 29-38. doi:10.1001/archneurol.2011.233.
For TBI/PTSD
Reed, J. R. (2017). Intranasal insulin in treating cortical and hippocampal injury after moderate TBI. Uniformed Services University.
Frey, W. H. (2013). Intranasal insulin to treat and protect against posttraumatic stress disorder. The Journal of nervous and mental disease,201(7), 638-639.
Brabazon, F. P., Khayrullina, G. I., Frey, W. H., & Byrnes, K. R. (2014, June). INTRANASAL INSULIN TREATMENT OF TRAUMATIC BRAIN INJURY. In JOURNAL OF NEUROTRAUMA (Vol. 31, No. 12, pp. A106-A106). 140 HUGUENOT STREET, 3RD FL, NEW ROCHELLE, NY 10801 USA: MARY ANN LIEBERT, INC.
IN Glutathione and Parkinson’s
Mischley, L. K., Conley, K. E., Shankland, E. G., Kavanagh, T. J., Rosenfeld, M. E., Duda, J. E., … & Padowski, J. M. (2016). Central nervous system uptake of intranasal glutathione in Parkinson’s disease. npj Parkinson’s Disease, 2, 16002.
Multimodal treatment IN Insulin/PRP/nutrients